Know the Rate-limiting Step in Glycolysis and How Pathway Keeps Going if You Run Out of Nad
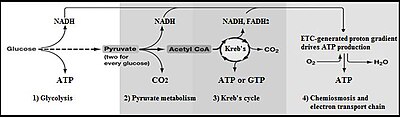
Summary of aerobic respiration
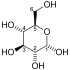
Glycolysis is the metabolic pathway that converts glucose C6H12O6, into pyruvic acid, CH3COCOOH. The free free energy released in this process is used to grade the high-free energy molecules adenosine triphosphate (ATP) and reduced nicotinamide adenine dinucleotide (NADH).[1] Glycolysis is a sequence of x reactions catalyzed by enzymes.
Glycolysis is a metabolic pathway that does non require oxygen. The broad occurrence of glycolysis in other species indicates that information technology is an ancient metabolic pathway.[2] Indeed, the reactions that brand up glycolysis and its parallel pathway, the pentose phosphate pathway, occur in the oxygen-free weather of the Archean oceans, also in the absenteeism of enzymes, catalyzed by metal.[3]
In well-nigh organisms, glycolysis occurs in the liquid office of cells, the cytosol. The most common type of glycolysis is the Embden–Meyerhof–Parnas (EMP) pathway, which was discovered by Gustav Embden, Otto Meyerhof, and Jakub Karol Parnas. Glycolysis also refers to other pathways, such as the Entner–Doudoroff pathway and various heterofermentative and homofermentative pathways. However, the discussion hither volition be express to the Embden–Meyerhof–Parnas pathway.[4]
The glycolysis pathway can be separated into two phases:[v]
- Investment phase – wherein ATP is consumed
- Yield stage – wherein more ATP is produced than originally consumed
Overview [edit]
The overall reaction of glycolysis is:

+ 2 [NAD]+
+ 2 [ADP]
+ 2 [P]i
ii × 
+ 2 [NADH]
+ 2 H+
+ two [ATP]
+ 2 H2O

Glycolysis pathway overview.
The utilise of symbols in this equation makes it appear unbalanced with respect to oxygen atoms, hydrogen atoms, and charges. Atom residue is maintained past the 2 phosphate (Pi) groups:[6]
- Each exists in the grade of a hydrogen phosphate anion (HPO4 2−), dissociating to contribute two H+ overall
- Each liberates an oxygen atom when it binds to an adenosine diphosphate (ADP) molecule, contributing iiO overall
Charges are balanced past the departure between ADP and ATP. In the cellular environs, all iii hydroxyl groups of ADP dissociate into −O− and H+, giving ADP3−, and this ion tends to exist in an ionic bond with Mg2+, giving ADPMg−. ATP behaves identically except that it has four hydroxyl groups, giving ATPMg2−. When these differences forth with the true charges on the two phosphate groups are considered together, the cyberspace charges of −iv on each side are balanced.
For simple fermentations, the metabolism of one molecule of glucose to ii molecules of pyruvate has a net yield of ii molecules of ATP. Nigh cells volition and so carry out further reactions to "repay" the used NAD+ and produce a final product of ethanol or lactic acid. Many bacteria use inorganic compounds every bit hydrogen acceptors to regenerate the NAD+.
Cells performing aerobic respiration synthesize much more than ATP, merely not as function of glycolysis. These further aerobic reactions utilize pyruvate, and NADH + H+ from glycolysis. Eukaryotic aerobic respiration produces approximately 34 additional molecules of ATP for each glucose molecule, however most of these are produced past a machinery vastly different from the substrate-level phosphorylation in glycolysis.
The lower-energy product, per glucose, of anaerobic respiration relative to aerobic respiration, results in greater flux through the pathway under hypoxic (low-oxygen) conditions, unless alternative sources of anaerobically oxidizable substrates, such as fat acids, are found.
| Metabolism of common monosaccharides, including glycolysis, gluconeogenesis, glycogenesis and glycogenolysis |
|---|
| |
History [edit]
The pathway of glycolysis as information technology is known today took almost 100 years to fully elucidate.[7] The combined results of many smaller experiments were required in club to empathise the pathway as a whole.
The first steps in understanding glycolysis began in the nineteenth century with the vino industry. For economic reasons, the French wine industry sought to investigate why wine sometimes turned distasteful, instead of fermenting into alcohol. French scientist Louis Pasteur researched this issue during the 1850s, and the results of his experiments began the long road to elucidating the pathway of glycolysis.[8] His experiments showed that fermentation occurs by the action of living microorganisms, yeasts, and that yeast'south glucose consumption decreased under aerobic weather condition of fermentation, in comparison to anaerobic conditions (the Pasteur effect).[9]

Eduard Buchner. Discovered prison cell-complimentary fermentation.
Insight into the component steps of glycolysis were provided by the non-cellular fermentation experiments of Eduard Buchner during the 1890s.[ten] [xi] Buchner demonstrated that the conversion of glucose to ethanol was possible using a not-living excerpt of yeast, due to the action of enzymes in the extract.[12] This experiment non only revolutionized biochemistry, just besides allowed later scientists to analyze this pathway in a more controlled laboratory setting. In a serial of experiments (1905-1911), scientists Arthur Harden and William Young discovered more pieces of glycolysis.[13] They discovered the regulatory furnishings of ATP on glucose consumption during alcohol fermentation. They also shed calorie-free on the role of one chemical compound as a glycolysis intermediate: fructose i,vi-bisphosphate.[14]
The elucidation of fructose ane,6-bisphosphate was accomplished by measuring COtwo levels when yeast juice was incubated with glucose. COtwo production increased speedily then slowed down. Harden and Immature noted that this process would restart if an inorganic phosphate (Pi) was added to the mixture. Harden and Young deduced that this process produced organic phosphate esters, and further experiments immune them to extract fructose diphosphate (F-one,vi-DP).
Arthur Harden and William Young along with Nick Sheppard determined, in a second experiment, that a heat-sensitive high-molecular-weight subcellular fraction (the enzymes) and a oestrus-insensitive low-molecular-weight cytoplasm fraction (ADP, ATP and NAD+ and other cofactors) are required together for fermentation to proceed. This experiment begun by observing that dialyzed (purified) yeast juice could non ferment or even create a sugar phosphate. This mixture was rescued with the addition of undialyzed yeast extract that had been boiled. Humid the yeast extract renders all proteins inactive (as it denatures them). The ability of boiled extract plus dialyzed juice to complete fermentation suggests that the cofactors were non-protein in character.[13]

Otto Meyerhof. One of the primary scientists involved in completing the puzzle of glycolysis
In the 1920s Otto Meyerhof was able to link together some of the many individual pieces of glycolysis discovered by Buchner, Harden, and Young. Meyerhof and his team were able to excerpt different glycolytic enzymes from muscle tissue, and combine them to artificially create the pathway from glycogen to lactic acrid.[xv] [sixteen]
In one paper, Meyerhof and scientist Renate Junowicz-Kockolaty investigated the reaction that splits fructose ane,6-diphosphate into the two triose phosphates. Previous work proposed that the split occurred via ane,3-diphosphoglyceraldehyde plus an oxidizing enzyme and cozymase. Meyerhoff and Junowicz constitute that the equilibrium constant for the isomerase and aldoses reaction were not affected by inorganic phosphates or any other cozymase or oxidizing enzymes. They further removed diphosphoglyceraldehyde equally a possible intermediate in glycolysis.[16]
With all of these pieces available by the 1930s, Gustav Embden proposed a detailed, step-by-step outline of that pathway we at present know as glycolysis.[17] The biggest difficulties in determining the intricacies of the pathway were due to the very short lifetime and depression steady-state concentrations of the intermediates of the fast glycolytic reactions. By the 1940s, Meyerhof, Embden and many other biochemists had finally completed the puzzle of glycolysis.[16] The agreement of the isolated pathway has been expanded in the subsequent decades, to include further details of its regulation and integration with other metabolic pathways.
Sequence of reactions [edit]
Summary of reactions [edit]
![]()

![]()
![]()

![]()

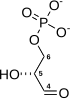
![]()
2 × 
![]()
two × 
![]()
2 × 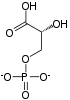
![]()
2 × 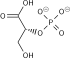
![]()
2 × 
![]()
2 × ![]()
Preparatory phase [edit]
The first five steps of Glycolysis are regarded equally the preparatory (or investment) phase, since they consume energy to convert the glucose into 2 iii-carbon sugar phosphates[5] (G3P).
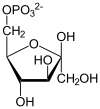
The first step is phosphorylation of glucose by a family unit of enzymes called hexokinases to form glucose 6-phosphate (G6P). This reaction consumes ATP, simply it acts to keep the glucose concentration low, promoting continuous transport of glucose into the jail cell through the plasma membrane transporters. In add-on, it blocks the glucose from leaking out – the prison cell lacks transporters for G6P, and complimentary improvidence out of the cell is prevented due to the charged nature of G6P. Glucose may alternatively be formed from the phosphorolysis or hydrolysis of intracellular starch or glycogen.
In animals, an isozyme of hexokinase called glucokinase is also used in the liver, which has a much lower analogousness for glucose (Km in the vicinity of normal glycemia), and differs in regulatory backdrop. The dissimilar substrate analogousness and alternate regulation of this enzyme are a reflection of the function of the liver in maintaining blood saccharide levels.
Cofactors: Mg2+
G6P is then rearranged into fructose 6-phosphate (F6P) by glucose phosphate isomerase. Fructose can likewise enter the glycolytic pathway by phosphorylation at this bespeak.
The change in structure is an isomerization, in which the G6P has been converted to F6P. The reaction requires an enzyme, phosphoglucose isomerase, to proceed. This reaction is freely reversible under normal cell conditions. However, it is often driven frontwards because of a low concentration of F6P, which is constantly consumed during the adjacent step of glycolysis. Under conditions of high F6P concentration, this reaction readily runs in reverse. This miracle tin be explained through Le Chatelier's Principle. Isomerization to a keto sugar is necessary for carbanion stabilization in the fourth reaction step (below).
The free energy expenditure of another ATP in this footstep is justified in 2 ways: The glycolytic procedure (up to this footstep) becomes irreversible, and the energy supplied destabilizes the molecule. Because the reaction catalyzed by phosphofructokinase 1 (PFK-1) is coupled to the hydrolysis of ATP (an energetically favorable step) information technology is, in essence, irreversible, and a different pathway must be used to exercise the reverse conversion during gluconeogenesis. This makes the reaction a cardinal regulatory indicate (encounter below). This is also the rate-limiting step.
Furthermore, the 2d phosphorylation event is necessary to allow the formation of two charged groups (rather than but i) in the subsequent step of glycolysis, ensuring the prevention of free improvidence of substrates out of the jail cell.
The same reaction tin also be catalyzed by pyrophosphate-dependent phosphofructokinase (PFP or PPi-PFK), which is found in most plants, some leaner, archea, and protists, merely non in animals. This enzyme uses pyrophosphate (PPi) as a phosphate donor instead of ATP. It is a reversible reaction, increasing the flexibility of glycolytic metabolism.[xviii] A rarer ADP-dependent PFK enzyme variant has been identified in archaean species.[19]
Cofactors: Mgtwo+
Destabilizing the molecule in the previous reaction allows the hexose band to exist dissever by aldolase into ii triose sugars: dihydroxyacetone phosphate (a ketose), and glyceraldehyde 3-phosphate (an aldose). There are two classes of aldolases: class I aldolases, nowadays in animals and plants, and class 2 aldolases, present in fungi and bacteria; the two classes use unlike mechanisms in cleaving the ketose ring.
Electrons delocalized in the carbon-carbon bond cleavage associate with the alcohol grouping. The resulting carbanion is stabilized by the structure of the carbanion itself via resonance charge distribution and past the presence of a charged ion prosthetic group.
Triosephosphate isomerase quickly interconverts dihydroxyacetone phosphate with glyceraldehyde 3-phosphate (GADP) that proceeds farther into glycolysis. This is advantageous, as information technology directs dihydroxyacetone phosphate down the aforementioned pathway as glyceraldehyde 3-phosphate, simplifying regulation.
Pay-off phase [edit]
The 2d half of glycolysis is known as the pay-off phase, characterised by a net gain of the energy-rich molecules ATP and NADH.[5] Since glucose leads to two triose sugars in the preparatory phase, each reaction in the pay-off stage occurs twice per glucose molecule. This yields 2 NADH molecules and iv ATP molecules, leading to a cyberspace gain of two NADH molecules and two ATP molecules from the glycolytic pathway per glucose.
The aldehyde groups of the triose sugars are oxidised, and inorganic phosphate is added to them, forming 1,3-bisphosphoglycerate.
The hydrogen is used to reduce two molecules of NAD+, a hydrogen carrier, to give NADH + H+ for each triose.
Hydrogen atom balance and charge balance are both maintained because the phosphate (Pi) group really exists in the form of a hydrogen phosphate anion (HPO4 two−),[6] which dissociates to contribute the extra H+ ion and gives a cyberspace charge of -3 on both sides.
Here, arsenate (AsOfour 3−), an anion akin to inorganic phosphate may replace phosphate as a substrate to course 1-arseno-three-phosphoglycerate. This, however, is unstable and readily hydrolyzes to form 3-phosphoglycerate, the intermediate in the next stride of the pathway. As a consequence of bypassing this step, the molecule of ATP generated from 1-3 bisphosphoglycerate in the adjacent reaction will not be made, even though the reaction proceeds. As a issue, arsenate is an uncoupler of glycolysis.[20]
This stride is the enzymatic transfer of a phosphate group from 1,3-bisphosphoglycerate to ADP by phosphoglycerate kinase, forming ATP and 3-phosphoglycerate. At this pace, glycolysis has reached the suspension-even point: 2 molecules of ATP were consumed, and 2 new molecules take now been synthesized. This pace, one of the two substrate-level phosphorylation steps, requires ADP; thus, when the prison cell has enough of ATP (and piddling ADP), this reaction does not occur. Because ATP decays relatively rapidly when it is not metabolized, this is an important regulatory point in the glycolytic pathway.
ADP actually exists as ADPMg−, and ATP as ATPMg2−, balancing the charges at −5 both sides.
Cofactors: Mg2+
Phosphoglycerate mutase isomerises 3-phosphoglycerate into 2-phosphoglycerate.
Enolase next converts 2-phosphoglycerate to phosphoenolpyruvate. This reaction is an emptying reaction involving an E1cB mechanism.
Cofactors: ii Mg2+, i "conformational" ion to coordinate with the carboxylate group of the substrate, and 1 "catalytic" ion that participates in the dehydration.
A last substrate-level phosphorylation now forms a molecule of pyruvate and a molecule of ATP by ways of the enzyme pyruvate kinase. This serves as an additional regulatory step, similar to the phosphoglycerate kinase step.
Cofactors: Mgtwo+
Biochemical logic [edit]
The existence of more than 1 betoken of regulation indicates that intermediates betwixt those points enter and leave the glycolysis pathway by other processes. For example, in the get-go regulated pace, hexokinase converts glucose into glucose-6-phosphate. Instead of standing through the glycolysis pathway, this intermediate tin can be converted into glucose storage molecules, such as glycogen or starch. The reverse reaction, breaking downwards, east.g., glycogen, produces mainly glucose-six-phosphate; very little gratis glucose is formed in the reaction. The glucose-6-phosphate so produced can enter glycolysis afterward the first command point.
In the 2nd regulated step (the 3rd footstep of glycolysis), phosphofructokinase converts fructose-vi-phosphate into fructose-1,6-bisphosphate, which then is converted into glyceraldehyde-three-phosphate and dihydroxyacetone phosphate. The dihydroxyacetone phosphate tin be removed from glycolysis by conversion into glycerol-3-phosphate, which can be used to class triglycerides.[21] Conversely, triglycerides can be broken down into fatty acids and glycerol; the latter, in plow, can exist converted into dihydroxyacetone phosphate, which tin enter glycolysis afterward the second control point.
Free free energy changes [edit]
| Chemical compound | Concentration / mM |
|---|---|
| Glucose | 5.0 |
| Glucose-six-phosphate | 0.083 |
| Fructose-6-phosphate | 0.014 |
| Fructose-1,6-bisphosphate | 0.031 |
| Dihydroxyacetone phosphate | 0.xiv |
| Glyceraldehyde-3-phosphate | 0.019 |
| 1,3-Bisphosphoglycerate | 0.001 |
| 2,three-Bisphosphoglycerate | four.0 |
| 3-Phosphoglycerate | 0.12 |
| 2-Phosphoglycerate | 0.03 |
| Phosphoenolpyruvate | 0.023 |
| Pyruvate | 0.051 |
| ATP | ane.85 |
| ADP | 0.fourteen |
| Pi | ane.0 |
The change in free energy, ΔG, for each pace in the glycolysis pathway can be calculated using ΔG = ΔG°' + RTln Q, where Q is the reaction quotient. This requires knowing the concentrations of the metabolites. All of these values are available for erythrocytes, with the exception of the concentrations of NAD+ and NADH. The ratio of NAD+ to NADH in the cytoplasm is approximately 1000, which makes the oxidation of glyceraldehyde-3-phosphate (pace 6) more favourable.
Using the measured concentrations of each step, and the standard gratuitous energy changes, the actual free energy change can be calculated. (Neglecting this is very common - the delta G of ATP hydrolysis in cells is non the standard gratuitous energy modify of ATP hydrolysis quoted in textbooks).
| Step | Reaction | ΔYard°' / (kJ/mol) | ΔThou / (kJ/mol) |
|---|---|---|---|
| one | Glucose + ATP4− → Glucose-6-phosphate2− + ADP3− + H+ | −16.vii | −34 |
| 2 | Glucose-6-phosphate2− → Fructose-6-phosphate2− | 1.67 | −ii.9 |
| 3 | Fructose-6-phosphate2− + ATP4− → Fructose-1,vi-bisphosphate4− + ADP3− + H+ | −xiv.2 | −19 |
| 4 | Fructose-i,six-bisphosphate4− → Dihydroxyacetone phosphate2− + Glyceraldehyde-3-phosphateii− | 23.ix | −0.23 |
| 5 | Dihydroxyacetone phosphateii− → Glyceraldehyde-3-phosphate2− | 7.56 | two.4 |
| 6 | Glyceraldehyde-3-phosphate2− + Pi two− + NAD+ → 1,3-Bisphosphoglycerate4− + NADH + H+ | 6.xxx | −i.29 |
| vii | ane,3-Bisphosphoglycerate4− + ADP3− → 3-Phosphoglycerate3− + ATP4− | −18.9 | 0.09 |
| 8 | 3-Phosphoglycerate3− → ii-Phosphoglycerate3− | iv.4 | 0.83 |
| 9 | 2-Phosphoglycerate3− → Phosphoenolpyruvate3− + H2O | 1.8 | 1.ane |
| 10 | Phosphoenolpyruvate3− + ADP3− + H+ → Pyruvate− + ATP4− | −31.seven | −23.0 |
From measuring the physiological concentrations of metabolites in an erythrocyte it seems that virtually 7 of the steps in glycolysis are in equilibrium for that cell type. Three of the steps — the ones with large negative free free energy changes — are non in equilibrium and are referred to as irreversible; such steps are often subject to regulation.
Step 5 in the effigy is shown backside the other steps, considering that step is a side-reaction that can decrease or increase the concentration of the intermediate glyceraldehyde-three-phosphate. That chemical compound is converted to dihydroxyacetone phosphate past the enzyme triose phosphate isomerase, which is a catalytically perfect enzyme; its charge per unit is and then fast that the reaction can be assumed to exist in equilibrium. The fact that ΔK is not zero indicates that the actual concentrations in the erythrocyte are not accurately known.
Regulation [edit]
Enzymes are the main components which drive the metabolic pathway and hence, exploring the regulatory mechanisms on these enzymes volition requite us insights to the regulatory processes affecting glycolysis. There are in total 9 primary steps in glycolysis which is driven by xiv different enzymes.[24] Enzymes can exist modified or are affected using 5 master regulatory processes including mail service-translational modification (PTM) and localization.
Biological mechanisms by which enzymes are regulated [edit]
ane. Cistron Expression
2. Allostery
3. Protein-protein interaction (PPI)
4. Post translational modification (PTM)
5. Localization
Regulation by insulin in animals [edit]
In animals, regulation of blood glucose levels by the pancreas in conjunction with the liver is a vital part of homeostasis. The beta cells in the pancreatic islets are sensitive to the blood glucose concentration.[25] A rise in the blood glucose concentration causes them to release insulin into the blood, which has an consequence especially on the liver, but too on fat and muscle cells, causing these tissues to remove glucose from the blood. When the blood sugar falls the pancreatic beta cells stop insulin production, but, instead, stimulate the neighboring pancreatic alpha cells to release glucagon into the blood.[25] This, in turn, causes the liver to release glucose into the blood past breaking down stored glycogen, and by means of gluconeogenesis. If the fall in the claret glucose level is especially rapid or severe, other glucose sensors cause the release of epinephrine from the adrenal glands into the claret. This has the aforementioned action as glucagon on glucose metabolism, merely its event is more pronounced.[25] In the liver glucagon and epinephrine cause the phosphorylation of the fundamental, charge per unit limiting enzymes of glycolysis, fatty acid synthesis, cholesterol synthesis, gluconeogenesis, and glycogenolysis. Insulin has the reverse effect on these enzymes.[26] The phosphorylation and dephosphorylation of these enzymes (ultimately in response to the glucose level in the claret) is the dominant manner past which these pathways are controlled in the liver, fat, and muscle cells. Thus the phosphorylation of phosphofructokinase inhibits glycolysis, whereas its dephosphorylation through the action of insulin stimulates glycolysis.[26]
Regulation of the rate limiting enzymes [edit]
The three regulatory enzymes are hexokinase (or glucokinase in the liver), phosphofructokinase, and pyruvate kinase. The flux through the glycolytic pathway is adjusted in response to conditions both inside and outside the jail cell. The internal factors that regulate glycolysis do so primarily to provide ATP in adequate quantities for the prison cell'due south needs. The external factors act primarily on the liver, fat tissue, and muscles, which can remove large quantities of glucose from the blood subsequently meals (thus preventing hyperglycemia by storing the excess glucose as fatty or glycogen, depending on the tissue type). The liver is also capable of releasing glucose into the blood between meals, during fasting, and practise thus preventing hypoglycemia by means of glycogenolysis and gluconeogenesis. These latter reactions coincide with the halting of glycolysis in the liver.
In addition hexokinase and glucokinase deed independently of the hormonal effects equally controls at the entry points of glucose into the cells of different tissues. Hexokinase responds to the glucose-6-phosphate (G6P) level in the cell, or, in the example of glucokinase, to the blood carbohydrate level in the blood to impart entirely intracellular controls of the glycolytic pathway in different tissues (see beneath).[26]
When glucose has been converted into G6P by hexokinase or glucokinase, it can either exist converted to glucose-1-phosphate (G1P) for conversion to glycogen, or information technology is alternatively converted by glycolysis to pyruvate, which enters the mitochondrion where it is converted into acetyl-CoA and then into citrate. Excess citrate is exported from the mitochondrion back into the cytosol, where ATP citrate lyase regenerates acetyl-CoA and oxaloacetate (OAA). The acetyl-CoA is then used for fat acid synthesis and cholesterol synthesis, ii important ways of utilizing excess glucose when its concentration is high in blood. The rate limiting enzymes catalyzing these reactions perform these functions when they have been dephosphorylated through the action of insulin on the liver cells. Betwixt meals, during fasting, exercise or hypoglycemia, glucagon and epinephrine are released into the blood. This causes liver glycogen to be converted back to G6P, and then converted to glucose by the liver-specific enzyme glucose 6-phosphatase and released into the blood. Glucagon and epinephrine also stimulate gluconeogenesis, which coverts non-saccharide substrates into G6P, which joins the G6P derived from glycogen, or substitutes for information technology when the liver glycogen shop accept been depleted. This is critical for brain role, since the brain utilizes glucose as an free energy source under nigh conditions.[27] The simultaneously phosphorylation of, particularly, phosphofructokinase, only also, to a certain extent pyruvate kinase, prevents glycolysis occurring at the aforementioned time as gluconeogenesis and glycogenolysis.
Hexokinase and glucokinase [edit]

All cells incorporate the enzyme hexokinase, which catalyzes the conversion of glucose that has entered the cell into glucose-half dozen-phosphate (G6P). Since the cell membrane is impervious to G6P, hexokinase essentially acts to transport glucose into the cells from which information technology can then no longer escape. Hexokinase is inhibited past high levels of G6P in the jail cell. Thus the rate of entry of glucose into cells partially depends on how fast G6P tin be disposed of past glycolysis, and by glycogen synthesis (in the cells which store glycogen, namely liver and muscles).[26] [28]
Glucokinase, different hexokinase, is not inhibited past G6P. Information technology occurs in liver cells, and will merely phosphorylate the glucose entering the cell to form glucose-6-phosphate (G6P), when the glucose in the blood is abundant. This existence the first step in the glycolytic pathway in the liver, it therefore imparts an boosted layer of control of the glycolytic pathway in this organ.[26]
Phosphofructokinase [edit]
Phosphofructokinase is an important control indicate in the glycolytic pathway, since it is 1 of the irreversible steps and has central allosteric effectors, AMP and fructose ii,six-bisphosphate (F2,6BP).
Fructose two,vi-bisphosphate (F2,6BP) is a very stiff activator of phosphofructokinase (PFK-1) that is synthesized when F6P is phosphorylated by a 2d phosphofructokinase (PFK2). In the liver, when blood saccharide is low and glucagon elevates cAMP, PFK2 is phosphorylated by poly peptide kinase A. The phosphorylation inactivates PFK2, and some other domain on this poly peptide becomes active every bit fructose bisphosphatase-2, which converts F2,6BP back to F6P. Both glucagon and epinephrine crusade high levels of cAMP in the liver. The event of lower levels of liver fructose-two,half-dozen-bisphosphate is a decrease in activeness of phosphofructokinase and an increase in activity of fructose 1,6-bisphosphatase, so that gluconeogenesis (in essence, "glycolysis in reverse") is favored. This is consistent with the role of the liver in such situations, since the response of the liver to these hormones is to release glucose to the blood.
ATP competes with AMP for the allosteric effector site on the PFK enzyme. ATP concentrations in cells are much higher than those of AMP, typically 100-fold higher,[29] simply the concentration of ATP does not change more than than about 10% nether physiological weather, whereas a 10% driblet in ATP results in a 6-fold increment in AMP.[30] Thus, the relevance of ATP equally an allosteric effector is questionable. An increase in AMP is a consequence of a decrease in energy charge in the cell.
Citrate inhibits phosphofructokinase when tested in vitro by enhancing the inhibitory event of ATP. However, information technology is hundred-to-one that this is a meaningful effect in vivo, because citrate in the cytosol is utilized mainly for conversion to acetyl-CoA for fatty acid and cholesterol synthesis.
TIGAR, a p53 induced enzyme, is responsible for the regulation of phosphofructokinase and acts to protect confronting oxidative stress.[31] TIGAR is a single enzyme with dual office that regulates F2,6BP. Information technology tin behave as a phosphatase (fructuose-two,6-bisphosphatase) which cleaves the phosphate at carbon-2 producing F6P. It can too behave equally a kinase (PFK2) calculation a phosphate onto carbon-ii of F6P which produces F2,6BP. In humans, the TIGAR protein is encoded past C12orf5 gene. The TIGAR enzyme volition hinder the forward progression of glycolysis, by creating a build up of fructose-6-phosphate (F6P) which is isomerized into glucose-6-phosphate (G6P). The accumulation of G6P volition shunt carbons into the pentose phosphate pathway.[32] [33]
Pyruvate kinase [edit]

Pyruvate kinase enzyme catalyzes the last footstep of glycolysis, in which pyruvate and ATP are formed. Pyruvate kinase catalyzes the transfer of a phosphate group from phosphoenolpyruvate (PEP) to ADP, yielding one molecule of pyruvate and one molecule of ATP.
Liver pyruvate kinase is indirectly regulated by epinephrine and glucagon, through protein kinase A. This poly peptide kinase phosphorylates liver pyruvate kinase to conciliate it. Musculus pyruvate kinase is not inhibited past epinephrine activation of protein kinase A. Glucagon signals fasting (no glucose available). Thus, glycolysis is inhibited in the liver but unaffected in muscle when fasting. An increase in claret sugar leads to secretion of insulin, which activates phosphoprotein phosphatase I, leading to dephosphorylation and activation of pyruvate kinase. These controls prevent pyruvate kinase from being active at the same time every bit the enzymes that catalyze the contrary reaction (pyruvate carboxylase and phosphoenolpyruvate carboxykinase), preventing a futile cycle.
Post-glycolysis processes [edit]
The overall procedure of glycolysis is:
- Glucose + two NAD+ + 2 ADP + 2 Pi → 2 pyruvate + two NADH + two H+ + 2 ATP
If glycolysis were to continue indefinitely, all of the NAD+ would be used upwards, and glycolysis would stop. To permit glycolysis to proceed, organisms must be able to oxidize NADH back to NAD+. How this is performed depends on which external electron acceptor is bachelor.
Anoxic regeneration of NAD+ [ commendation needed ] [edit]
One method of doing this is to simply have the pyruvate do the oxidation; in this process, pyruvate is converted to lactate (the conjugate base of operations of lactic acrid) in a procedure called lactic acid fermentation:
- Pyruvate + NADH + H+ → lactate + NAD+
This process occurs in the bacteria involved in making yogurt (the lactic acid causes the milk to curdle). This process also occurs in animals under hypoxic (or partially anaerobic) atmospheric condition, found, for instance, in overworked muscles that are starved of oxygen. In many tissues, this is a cellular last resort for energy; nigh animal tissue cannot tolerate anaerobic conditions for an extended period of time.
Some organisms, such as yeast, catechumen NADH back to NAD+ in a process called ethanol fermentation. In this process, the pyruvate is converted showtime to acetaldehyde and carbon dioxide, and then to ethanol.
Lactic acrid fermentation and ethanol fermentation tin can occur in the absence of oxygen. This anaerobic fermentation allows many single-jail cell organisms to utilise glycolysis as their but free energy source.
Anoxic regeneration of NAD+ is only an effective means of energy production during short, intense exercise in vertebrates, for a period ranging from 10 seconds to two minutes during a maximal effort in humans. (At lower exercise intensities information technology can sustain musculus activity in diving animals, such every bit seals, whales and other aquatic vertebrates, for very much longer periods of time.) Under these weather NAD+ is replenished by NADH donating its electrons to pyruvate to form lactate. This produces ii ATP molecules per glucose molecule, or about 5% of glucose'due south energy potential (38 ATP molecules in leaner). Just the speed at which ATP is produced in this mode is virtually 100 times that of oxidative phosphorylation. The pH in the cytoplasm quickly drops when hydrogen ions accrue in the muscle, somewhen inhibiting the enzymes involved in glycolysis.
The burning awareness in muscles during difficult exercise tin can be attributed to the release of hydrogen ions during the shift to glucose fermentation from glucose oxidation to carbon dioxide and water, when aerobic metabolism can no longer keep pace with the energy demands of the muscles. These hydrogen ions form a part of lactic acrid. The body falls dorsum on this less efficient but faster method of producing ATP under depression oxygen atmospheric condition. This is thought to have been the main ways of free energy production in earlier organisms earlier oxygen reached loftier concentrations in the atmosphere between 2000 and 2500 million years ago, and thus would represent a more than ancient form of free energy production than the aerobic replenishment of NAD+ in cells.
The liver in mammals gets rid of this excess lactate by transforming information technology back into pyruvate nether aerobic conditions; see Cori bike.
Fermentation of pyruvate to lactate is sometimes also chosen "anaerobic glycolysis", even so, glycolysis ends with the production of pyruvate regardless of the presence or absence of oxygen.
In the above two examples of fermentation, NADH is oxidized by transferring two electrons to pyruvate. Still, anaerobic bacteria use a wide diversity of compounds as the terminal electron acceptors in cellular respiration: nitrogenous compounds, such as nitrates and nitrites; sulfur compounds, such as sulfates, sulfites, sulfur dioxide, and elemental sulfur; carbon dioxide; iron compounds; manganese compounds; cobalt compounds; and uranium compounds.
Aerobic regeneration of NAD+, and disposal of pyruvate [edit]
In aerobic organisms, a complex machinery has been adult to utilize the oxygen in air as the final electron acceptor.
- Firstly, the NADH + H+ generated past glycolysis has to be transferred to the mitochondrion to exist oxidized, and thus to regenerate the NAD+ necessary for glycolysis to continue. However the inner mitochondrial membrane is impermeable to NADH and NAD+.[34] Employ is therefore made of two "shuttles" to ship the electrons from NADH across the mitochondrial membrane. They are the malate-aspartate shuttle and the glycerol phosphate shuttle. In the former the electrons from NADH are transferred to cytosolic oxaloacetate to form malate. The malate then traverses the inner mitochondrial membrane into the mitochondrial matrix, where it is reoxidized by NAD+ forming intra-mitochondrial oxaloacetate and NADH. The oxaloacetate is and then re-cycled to the cytosol via its conversion to aspartate which is readily transported out of the mitochondrion. In the glycerol phosphate shuttle electrons from cytosolic NADH are transferred to dihydroxyacetone to form glycerol-iii-phosphate which readily traverses the outer mitochondrial membrane. Glycerol-3-phosphate is then reoxidized to dihydroxyacetone, donating its electrons to FAD instead of NAD+.[34] This reaction takes identify on the inner mitochondrial membrane, allowing FADH2 to donate its electrons directly to coenzyme Q (ubiquinone) which is function of the electron transport concatenation which ultimately transfers electrons to molecular oxygen (O2), with the formation of h2o, and the release of energy eventually captured in the course of ATP.
- The glycolytic end-production, pyruvate (plus NAD+) is converted to acetyl-CoA, CO2 and NADH + H+ within the mitochondria in a process chosen pyruvate decarboxylation.
- The resulting acetyl-CoA enters the citric acid bicycle (or Krebs Bicycle), where the acetyl group of the acetyl-CoA is converted into carbon dioxide by two decarboxylation reactions with the formation of yet more intra-mitochondrial NADH + H+.
- The intra-mitochondrial NADH + H+ is oxidized to NAD+ by the electron transport chain, using oxygen as the final electron acceptor to class h2o. The energy released during this process is used to create a hydrogen ion (or proton) gradient across the inner membrane of the mitochondrion.
- Finally, the proton gradient is used to produce about two.five ATP for every NADH + H+ oxidized in a process chosen oxidative phosphorylation.[34]
Conversion of carbohydrates into fatty acids and cholesterol [edit]
The pyruvate produced by glycolysis is an important intermediary in the conversion of carbohydrates into fatty acids and cholesterol.[35] This occurs via the conversion of pyruvate into acetyl-CoA in the mitochondrion. Still, this acetyl CoA needs to exist transported into cytosol where the synthesis of fatty acids and cholesterol occurs. This cannot occur directly. To obtain cytosolic acetyl-CoA, citrate (produced by the condensation of acetyl CoA with oxaloacetate) is removed from the citric acrid cycle and carried across the inner mitochondrial membrane into the cytosol.[35] There it is broken past ATP citrate lyase into acetyl-CoA and oxaloacetate. The oxaloacetate is returned to mitochondrion as malate (and then back into oxaloacetate to transfer more acetyl-CoA out of the mitochondrion). The cytosolic acetyl-CoA can exist carboxylated by acetyl-CoA carboxylase into malonyl CoA, the first committed step in the synthesis of fatty acids, or it can be combined with acetoacetyl-CoA to course three-hydroxy-iii-methylglutaryl-CoA (HMG-CoA) which is the rate limiting step controlling the synthesis of cholesterol.[36] Cholesterol can be used as is, as a structural component of cellular membranes, or it can exist used to synthesize the steroid hormones, bile salts, and vitamin D.[28] [35] [36]
Conversion of pyruvate into oxaloacetate for the citric acrid cycle [edit]
Pyruvate molecules produced by glycolysis are actively transported beyond the inner mitochondrial membrane, and into the matrix where they can either be oxidized and combined with coenzyme A to form CO2, acetyl-CoA, and NADH,[28] or they can exist carboxylated (past pyruvate carboxylase) to course oxaloacetate. This latter reaction "fills up" the amount of oxaloacetate in the citric acrid wheel, and is therefore an anaplerotic reaction (from the Greek significant to "fill up"), increasing the cycle's capacity to metabolize acetyl-CoA when the tissue'south energy needs (e.m. in middle and skeletal muscle) are suddenly increased by activity.[37] In the citric acid cycle all the intermediates (e.g. citrate, iso-citrate, alpha-ketoglutarate, succinate, fumarate, malate and oxaloacetate) are regenerated during each plough of the cycle. Calculation more than of any of these intermediates to the mitochondrion therefore means that that additional amount is retained within the wheel, increasing all the other intermediates as one is converted into the other. Hence the addition of oxaloacetate greatly increases the amounts of all the citric acid intermediates, thereby increasing the cycle'south capacity to metabolize acetyl CoA, converting its acetate component into CO2 and water, with the release of plenty energy to class 11 ATP and 1 GTP molecule for each additional molecule of acetyl CoA that combines with oxaloacetate in the bicycle.[37]
To cataplerotically remove oxaloacetate from the citric cycle, malate tin can exist transported from the mitochondrion into the cytoplasm, decreasing the amount of oxaloacetate that tin be regenerated.[37] Furthermore, citric acid intermediates are constantly used to form a variety of substances such as the purines, pyrimidines and porphyrins.[37]
Intermediates for other pathways [edit]
This article concentrates on the catabolic role of glycolysis with regard to converting potential chemical energy to usable chemic energy during the oxidation of glucose to pyruvate. Many of the metabolites in the glycolytic pathway are as well used by anabolic pathways, and, as a consequence, flux through the pathway is disquisitional to maintain a supply of carbon skeletons for biosynthesis.
The post-obit metabolic pathways are all strongly reliant on glycolysis as a source of metabolites: and many more.
- Pentose phosphate pathway, which begins with the dehydrogenation of glucose-6-phosphate, the first intermediate to be produced by glycolysis, produces various pentose sugars, and NADPH for the synthesis of fatty acids and cholesterol.
- Glycogen synthesis also starts with glucose-half dozen-phosphate at the get-go of the glycolytic pathway.
- Glycerol, for the formation of triglycerides and phospholipids, is produced from the glycolytic intermediate glyceraldehyde-three-phosphate.
- Various post-glycolytic pathways:
-
- Fat acrid synthesis
- Cholesterol synthesis
- The citric acid cycle which in turn leads to:
-
- Amino acid synthesis
- Nucleotide synthesis
- Tetrapyrrole synthesis
Although gluconeogenesis and glycolysis share many intermediates the one is not functionally a branch or tributary of the other. There are 2 regulatory steps in both pathways which, when active in the one pathway, are automatically inactive in the other. The two processes tin therefore not be simultaneously active.[38] Indeed, if both sets of reactions were highly active at the aforementioned time the net result would be the hydrolysis of four high free energy phosphate bonds (two ATP and two GTP) per reaction bicycle.[38]
NAD+ is the oxidizing agent in glycolysis, every bit information technology is in near other energy yielding metabolic reactions (e.yard. beta-oxidation of fatty acids, and during the citric acid cycle). The NADH thus produced is primarily used to ultimately transfer electrons to O2 to produce h2o, or, when O2 is non available, to produced compounds such every bit lactate or ethanol (see Anoxic regeneration of NAD+ to a higher place). NADH is rarely used for synthetic processes, the notable exception being gluconeogenesis. During fatty acid and cholesterol synthesis the reducing amanuensis is NADPH. This difference exemplifies a general principle that NADPH is consumed during biosynthetic reactions, whereas NADH is generated in energy-yielding reactions.[38] The source of the NADPH is ii-fold. When malate is oxidatively decarboxylated by "NADP+-linked malic enzyme" pyruvate, CO2 and NADPH are formed. NADPH is also formed by the pentose phosphate pathway which converts glucose into ribose, which can be used in synthesis of nucleotides and nucleic acids, or information technology can exist catabolized to pyruvate.[38]
Glycolysis in disease [edit]
Diabetes [edit]
Cellular uptake of glucose occurs in response to insulin signals, and glucose is subsequently broken downwardly through glycolysis, lowering blood sugar levels. However, the low insulin levels seen in diabetes result in hyperglycemia, where glucose levels in the blood rise and glucose is not properly taken upwardly by cells. Hepatocytes farther contribute to this hyperglycemia through gluconeogenesis. Glycolysis in hepatocytes controls hepatic glucose production, and when glucose is overproduced by the liver without having a means of being broken down by the body, hyperglycemia results.[39]
Genetic diseases [edit]
Glycolytic mutations are by and large rare due to importance of the metabolic pathway, this means that the majority of occurring mutations consequence in an inability for the cell to respire, and therefore cause the expiry of the cell at an early phase. However, some mutations are seen with one notable example being Pyruvate kinase deficiency, leading to chronic hemolytic anemia.
Cancer [edit]
Cancerous tumor cells perform glycolysis at a charge per unit that is ten times faster than their noncancerous tissue counterparts.[forty] During their genesis, limited capillary support frequently results in hypoxia (decreased O2 supply) inside the tumor cells. Thus, these cells rely on anaerobic metabolic processes such as glycolysis for ATP (adenosine triphosphate). Some tumor cells overexpress specific glycolytic enzymes which issue in higher rates of glycolysis.[41] Often these enzymes are Isoenzymes, of traditional glycolysis enzymes, that vary in their susceptibility to traditional feedback inhibition. The increase in glycolytic activity ultimately counteracts the furnishings of hypoxia by generating sufficient ATP from this anaerobic pathway.[42] This phenomenon was get-go described in 1930 by Otto Warburg and is referred to as the Warburg event. The Warburg hypothesis claims that cancer is primarily caused past dysfunctionality in mitochondrial metabolism, rather than because of the uncontrolled growth of cells. A number of theories accept been avant-garde to explain the Warburg effect. Ane such theory suggests that the increased glycolysis is a normal protective procedure of the body and that cancerous change could be primarily caused past free energy metabolism.[43]
This high glycolysis rate has important medical applications, as high aerobic glycolysis by malignant tumors is utilized clinically to diagnose and monitor treatment responses of cancers by imaging uptake of 2-18F-2-deoxyglucose (FDG) (a radioactive modified hexokinase substrate) with positron emission tomography (PET).[44] [45]
There is ongoing inquiry to affect mitochondrial metabolism and care for cancer by reducing glycolysis and thus starving malignant cells in various new means, including a ketogenic diet.[46] [47] [48]
Interactive pathway map [edit]
The diagram below shows human protein names. Names in other organisms may be different and the number of isozymes (such as HK1, HK2, ...) is likely to exist different also.
Click on genes, proteins and metabolites beneath to link to corresponding articles. [§ one]
[[File:

[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]
[[]]

|alt=Glycolysis and Gluconeogenesis edit]]
Glycolysis and Gluconeogenesis edit
- ^ The interactive pathway map tin can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
Alternative classification [edit]
Some of the metabolites in glycolysis have alternative names and classification. In part, this is because some of them are common to other pathways, such as the Calvin cycle.
| This article | Alternative | |||
|---|---|---|---|---|
| one | Glucose | Glc | Dextrose | |
| 2 | Glucose-6-phosphate | G6P | ||
| iii | Fructose-6-phosphate | F6P | ||
| four | Fructose-1,half dozen-bisphosphate | F1,6BP | Fructose one,6-diphosphate | FBP; FDP; F1,6DP |
| five | Dihydroxyacetone phosphate | DHAP | Glycerone phosphate | |
| six | Glyceraldehyde-3-phosphate | GADP | iii-Phosphoglyceraldehyde | PGAL; G3P; GALP; GAP; TP |
| 7 | 1,three-Bisphosphoglycerate | i,3BPG | Glycerate-1,3-bisphosphate, glycerate-1,3-diphosphate, ane,3-diphosphoglycerate | PGAP; BPG; DPG |
| 8 | 3-Phosphoglycerate | 3PG | Glycerate-iii-phosphate | PGA; GP |
| ix | two-Phosphoglycerate | 2PG | Glycerate-2-phosphate | |
| 10 | Phosphoenolpyruvate | PEP | ||
| 11 | Pyruvate | Pyr | Pyruvic acid | |
Construction of glycolysis components in Fischer projections and polygonal model [edit]
The intermediates of glycolysis depicted in Fischer projections prove the chemical changing step by pace. Such image can be compared to polygonal model representation.[49] Some other comparation of Fischer projections and Poligonal Model in glycolysis is shown in a video.[50] Video animations in the aforementioned channel in YouTube can be seen for another metabolic pathway (Krebs Bike) and the representation and applying of Polygonal Model in Organic Chemical science [51]

Glycolysis - Construction of anaerobic glycolysis components showed using Fischer projections, left, and polygonal model, right. The compounds correspond to glucose (GLU), glucose vi-phosphate (G6P), fructose 6-phosphate (F6P), fructose 1,six-bisphosphate ( F16BP), dihydroxyacetone phosphate (DHAP), glyceraldehyde 3-phosphate(GA3P), 1,3-bisphosphoglycerate (13BPG), 3-phosphoglycerate (3PG), ii-phosphoglycerate (2PG), phosphoenolpyruvate (PEP), pyruvate (PIR), and lactate (LAC). The enzymes which participate of this pathway are indicated past underlined numbers, and represent to hexokinase (
1), glucose-6-phosphate isomerase (
2), phosphofructokinase-1 (
three), fructose-bisphosphate aldolase (
four), triosephosphate isomerase (
v), glyceraldehyde-3-phosphate dehydrogenase (
5), phosphoglycerate kinase (
vii), phosphoglycerate mutase (
8), phosphopyruvate hydratase (enolase) (
9), pyruvate kinase (
10), and lactate dehydrogenase (
xi). The participant coenzymes (NAD+, NADH + H+, ATP and ADP), inorganic phosphate, H2O and CO2 were omitted in these representations. The phosphorylation reactions from ATP, also the ADP phosphorylation reactions in after steps of glycolysis are shown as ~P respectively entering or going out the pathway. The oxireduction reactions using NAD+ or NADH are observed as hydrogens "2H" going out or inbound the pathway.
Encounter also [edit]
| | Wikimedia Commons has media related to Glycolysis. |
- Sugar catabolism
- Citric acid cycle
- Cori cycle
- Fermentation (biochemistry)
- Gluconeogenesis
- Glycolytic oscillation
- Pentose phosphate pathway
- Pyruvate decarboxylation
- Triose kinase
References [edit]
- ^ Alfarouk, Khalid O.; Verduzco, Daniel; Rauch, Cyril; Muddathir, Abdel Khalig; Bashir, Adil H. H.; Elhassan, Gamal O.; Ibrahim, Muntaser Due east.; Orozco, Julian David Polo; Cardone, Rosa Angela; Reshkin, Stephan J.; Harguindey, Salvador (18 December 2014). "Glycolysis, tumor metabolism, cancer growth and dissemination. A new pH-based etiopathogenic perspective and therapeutic approach to an old cancer question". Oncoscience. 1 (12): 777–802. doi:10.18632/oncoscience.109.
- ^ Romano, AH; Conway, T (1996). "Evolution of saccharide metabolic pathways". Res Microbiol. 147 (half-dozen–vii): 448–55. doi:x.1016/0923-2508(96)83998-2. PMID 9084754.
- ^ Keller; Ralser; Turchyn (Apr 2014). "Not-enzymatic glycolysis and pentose phosphate pathway-similar reactions in a plausible Archean ocean". Mol Syst Biol. ten (four): 725. doi:x.1002/msb.20145228. PMC4023395. PMID 24771084.
- ^ Kim BH, Gadd GM. (2011) Bacterial Physiology and Metabolism, 3rd edition.
- ^ a b c Glycolysis – Blitheness and Notes
- ^ a b Lane, A. N.; Fan, T. W. -M.; Higashi, R. M. (2009). "Metabolic acidosis and the importance of balanced equations". Metabolomics. 5 (2): 163–165. doi:ten.1007/s11306-008-0142-2. S2CID 35500999.
- ^ Barnett JA (April 2003). "A history of inquiry on yeasts 5: the fermentation pathway". Yeast. 20 (6): 509–543. doi:10.1002/yea.986. PMID 12722184. S2CID 26805351.
- ^ "Louis Pasteur and Alcoholic Fermentation". www.pasteurbrewing.com. Archived from the original on 2011-01-thirteen. Retrieved 2016-02-23 .
- ^ "Yeast, Fermentation, Beer, Vino". world wide web.nature.com . Retrieved 2016-02-23 .
- ^ Kohler, Robert (1971-03-01). "The groundwork to Eduard Buchner'southward discovery of cell-free fermentation". Periodical of the History of Biology. iv (i): 35–61. doi:10.1007/BF00356976. ISSN 0022-5010. PMID 11609437. S2CID 46573308.
- ^ "Eduard Buchner - Biographical". world wide web.nobelprize.org . Retrieved 2016-02-23 .
- ^ Cornish-Bowden, Athel (1997). "Harden and Young's Discovery of Fructose 1,6-Bisphosphate". New Beer in an Old Canteen: Eduard Buchner and the Growth of Biochemical Knowledge. Valencia, Spain. pp. 135–148.
- ^ a b Palmer, Grahm. "Chapter 3". Bios 302.
- ^ Cornish-Bowden, Athel (1997). "Harden and Young's Discovery of Fructose 1,half dozen-Bisphosphate". New Beer in an Old Bottle: Eduard Buchner and the Growth of Biochemical Knowledge. Valencia, Spain. pp. 151–158.
- ^ "Otto Meyerhof - Biographical". www.nobelprize.org . Retrieved 2016-02-23 .
- ^ a b c Kresge, Nicole; Simoni, Robert D.; Colina, Robert L. (2005-01-28). "Otto Fritz Meyerhof and the Elucidation of the Glycolytic Pathway". Journal of Biological Chemistry. 280 (four): e3. doi:x.1016/S0021-9258(20)76366-0. ISSN 0021-9258. PMID 15665335.
- ^ "Embden, Gustav – Lexicon definition of Embden, Gustav | Encyclopedia.com: Gratis online dictionary". www.encyclopedia.com . Retrieved 2016-02-23 .
- ^ Reeves, R. East.; S D. J.; Blytt H. J.; Warren Fifty. M. (1974). "Pyrophosphate: D-fructose 6-phosphate one-phosphotransferase. A new enzyme with the glycolytic function half dozen-phosphate 1-phosphotransferase". J Biol Chem. 249 (24): 7737–7741. doi:10.1016/S0021-9258(xix)42029-2. PMID 4372217.
- ^ Selig, M.; Xavier K. B.; Santos H.; Schönheit P. (1997). "Comparative analysis of Embden-Meyerhof and Entner-Doudoroff glycolytic pathways in hyperthermophilic archaea and the bacterium Thermotoga". Curvation Microbiol. 167 (4): 217–232. doi:ten.1007/BF03356097. PMID 9075622. S2CID 19489719.
- ^ Garrett, Reginald H.; Grisham, Charles M. (2012). Biochemistry. Cengage Learning; 5 edition. ISBN978-one-133-10629-6.
- ^ Berg, J. Thousand.; Tymoczko, J. L.; Stryer, Fifty. (2007). Biochemistry (sixth ed.). New York: Freeman. p. 622. ISBN978-0716787242.
- ^ Garrett, R.; Grisham, C. One thousand. (2005). Biochemistry (tertiary ed.). Belmont, CA: Thomson Brooks/Cole. p. 584. ISBN978-0-534-49033-1.
- ^ Garrett, R.; Grisham, C. One thousand. (2005). Biochemistry (3rd ed.). Belmont, CA: Thomson Brooks/Cole. pp. 582–583. ISBN978-0-534-49033-1.
- ^ Hollinshead WD, Rodriguez S, Martin HG, Wang Yard, Baidoo EE, Sale KL, Keasling JD, Mukhopadhyay A, Tang YJ. Examining Escherichia coli glycolytic pathways, catabolite repression, and metabolite channeling using Δ pfk mutants. Biotechnology for biofuels. 2016 December;ix(i):i-3.
- ^ a b c Koeslag, Johan H.; Saunders, Peter T.; Terblanche, Elmarie (2003). "Topical Review: A reappraisal of the claret glucose homeostat which comprehensively explains the blazon ii diabetes-syndrome X circuitous". Journal of Physiology. 549 (Pt ii): 333–346. doi:10.1113/jphysiol.2002.037895. PMC2342944. PMID 12717005.
- ^ a b c d e Stryer, Lubert (1995). "Glycolysis.". In: Biochemistry (4th ed.). New York: Due west.H. Freeman and Visitor. pp. 483–508. ISBN0-7167-2009-4.
- ^ Stryer, Lubert (1995). Biochemistry (Fourth ed.). New York: W.H. Freeman and Company. p. 773. ISBN0-7167-2009-4.
- ^ a b c Voet, Donald; Judith G. Voet; Charlotte West. Pratt (2006). Fundamentals of Biochemistry, 2nd Edition. John Wiley and Sons, Inc. pp. 547, 556. ISBN978-0-471-21495-3.
- ^ Beis, I.; Newsholme, Due east. A. (1975). "The contents of adenine nucleotides, phosphagens and some glycolytic intermediates in resting muscles from vertebrates and invertebrates". Biochem J. 152 (1): 23–32. doi:10.1042/bj1520023. PMC1172435. PMID 1212224.
- ^ Voet D., and Voet J. G. (2004). Biochemistry 3rd Edition (New York, John Wiley & Sons, Inc.).
- ^ Lackie, John (2010). TIGAR. Oxford Reference Online: Oxford University Press. ISBN9780199549351.
- ^ Bensaad, Karim (July 16, 2006). "TIGAR, a p53-Inducible Regulator of Glycolysis and Apoptosis". Cell. 126 (I): 107–120. doi:10.1016/j.cell.2006.05.036. PMID 16839880. S2CID 15006256.
- ^ "TIGAR TP53 induced glycolysis regulatory phosphatase [Homo sapiens (human being)] - Gene - NCBI". world wide web.ncbi.nlm.nih.gov . Retrieved 2018-05-17 .
- ^ a b c Stryer, Lubert (1995). "Oxidative phosphorylation.". In: Biochemistry (Fourth ed.). New York: W.H. Freeman and Company. pp. 537–549. ISBN0-7167-2009-four.
- ^ a b c Stryer, Lubert (1995). "Fatty acid metabolism.". In: Biochemistry (Fourth ed.). New York: W.H. Freeman and Company. pp. 603–628. ISBN0-7167-2009-4.
- ^ a b Stryer, Lubert (1995). "Biosynthesis of membrane lipids and steroids.". In: Biochemistry (Quaternary ed.). New York: Westward.H. Freeman and Visitor. pp. 691–707. ISBN0-7167-2009-four.
- ^ a b c d Stryer, Lubert (1995). "Citric acrid bike.". In: Biochemistry (Fourth ed.). New York: Due west.H. Freeman and Company. pp. 509–527, 569–579, 614–616, 638–641, 732–735, 739–748, 770–773. ISBN0-7167-2009-4.
- ^ a b c d Stryer, Lubert (1995). Biochemistry (4th ed.). New York: W.H. Freeman and Company. pp. 559–565, 574–576, 614–623. ISBN0-7167-2009-4.
- ^ Guo, Xin; Li, Honggui; Xu, Hang; Woo, Shihlung; Dong, Hui; Lu, Fuer; Lange, Alex J.; Wu, Chaodong (2012-08-01). "Glycolysis in the control of blood glucose homeostasis". Acta Pharmaceutica Sinica B. 2 (4): 358–367. doi:ten.1016/j.apsb.2012.06.002. ISSN 2211-3835.
- ^ Alfarouk, KO; Verduzco, D; Rauch, C; Muddathir, AK; Adil, HH; Elhassan, GO; Ibrahim, ME; David Polo Orozco, J; Cardone, RA; Reshkin, SJ; Harguindey, S (2014). "Glycolysis, tumor metabolism, cancer growth and broadcasting. A new pH-based etiopathogenic perspective and therapeutic approach to an old cancer question". Oncoscience. 1 (12): 777–802. doi:x.18632/oncoscience.109. PMC4303887. PMID 25621294.
- ^ Alfarouk, KO; Shayoub, ME; Muddathir, AK; Elhassan, Go; Bashir, AH (22 July 2011). "Evolution of Tumor Metabolism might Reflect Carcinogenesis equally a Reverse Development process (Dismantling of Multicellularity)". Cancers. three (3): 3002–17. doi:10.3390/cancers3033002. PMC3759183. PMID 24310356.
- ^ Nelson, David 50.; Cox, Michael Chiliad. (2005). Lehninger principles of biochemistry (4th ed.). New York: West.H. Freeman. ISBN978-0-7167-4339-2.
- ^ Gold, Joseph (October 2011). "What is Cancer?". Archived from the original on May 19, 2018. Retrieved September 8, 2012.
- ^ "4320139 549..559" (PDF) . Retrieved Dec 5, 2005.
- ^ "PET Scan: PET Browse Info Reveals ..." Retrieved December 5, 2005.
- ^ Schwartz, L; Seyfried, T; Alfarouk, KO; Da Veiga Moreira, J; Fais, S (April 2017). "Out of Warburg outcome: An constructive cancer handling targeting the tumor specific metabolism and dysregulated pH". Seminars in Cancer Biology. 43: 134–138. doi:10.1016/j.semcancer.2017.01.005. PMID 28122260.
- ^ Schwartz, L; Supuran, CT; Alfarouk, KO (2017). "The Warburg Effect and the Hallmarks of Cancer". Anti-Cancer Agents in Medicinal Chemistry. 17 (2): 164–170. doi:10.2174/1871520616666161031143301. PMID 27804847.
- ^ Maroon, J; Bost J; Amos A; Zuccoli G (May 2013). "Restricted Calorie Ketogenic Diet for the Handling of Glioblastoma Multiforme". Journal of Child Neurology. 28 (8): 1002–1008. doi:10.1177/0883073813488670. PMID 23670248. S2CID 1994087.
- ^ Bonafe, C. F. S.; Bispo, J. A. C.; de Jesus, M. B. (2018). The Polygonal Model: A Simple Representation of Biomolecules equally a Tool for Teaching Metabolism. Biochemistry and Molecular Biology Educational activity. 46: 66-75. DOI - 10.1002/bmb.21093.
- ^ Bonafe, Carlos (23 September 2019). "Introduction to Polygonal Model - Function ane. Glycolysis and Structure of the Participant Molecules". YouTube. Archived from the original on 2021-xi-04.
- ^ "Metabolism Animation and Polygonal Model". YouTube . Retrieved 2019-12-xi .
External links [edit]
- A Detailed Glycolysis Blitheness provided by IUBMB (Adobe Flash Required)
- The Glycolytic enzymes in Glycolysis at RCSB PDB
- Glycolytic bicycle with animations at wdv.com
- Metabolism, Cellular Respiration and Photosynthesis - The Virtual Library of Biochemistry, Molecular Biology and Cell Biological science
- The chemical logic behind glycolysis at ufp.pt
- Expasy biochemical pathways poster at ExPASy
- MedicalMnemonics.com: 317 5468
- metpath: Interactive representation of glycolysis
Source: https://en.wikipedia.org/wiki/Glycolysis

0 Response to "Know the Rate-limiting Step in Glycolysis and How Pathway Keeps Going if You Run Out of Nad"
Post a Comment